Oxygen molecular orbital diagram Oxygen atom lewis structure O2 molecular orbital diagram o2 molecular orbital diagram vs lewis structure
SOLVED: SQ 1: Empty versions of the molecular orbital diagrams for O2
Single oxygen lewis structure Lewis structure of o2 (with 6 simple steps to draw!) O2 lewis structure & molecular geometry
Diatomic oxygen lewis structure
Orbital molecular he2 o2 be2 bonding paramagnetic diamagnetic orbitals electrons diatomic molecules chem unpaired labeled sigma antibonding inorganic mixing nitrogenOxygen molecular orbital diagram A lewis structure obeying the octet rule can be drawn for o2 as follows21+ o22- mo diagram.
O2 2 lewis structureSingle oxygen lewis structure O2 oxygen molecular hybridizationOrbital diagram for o2.
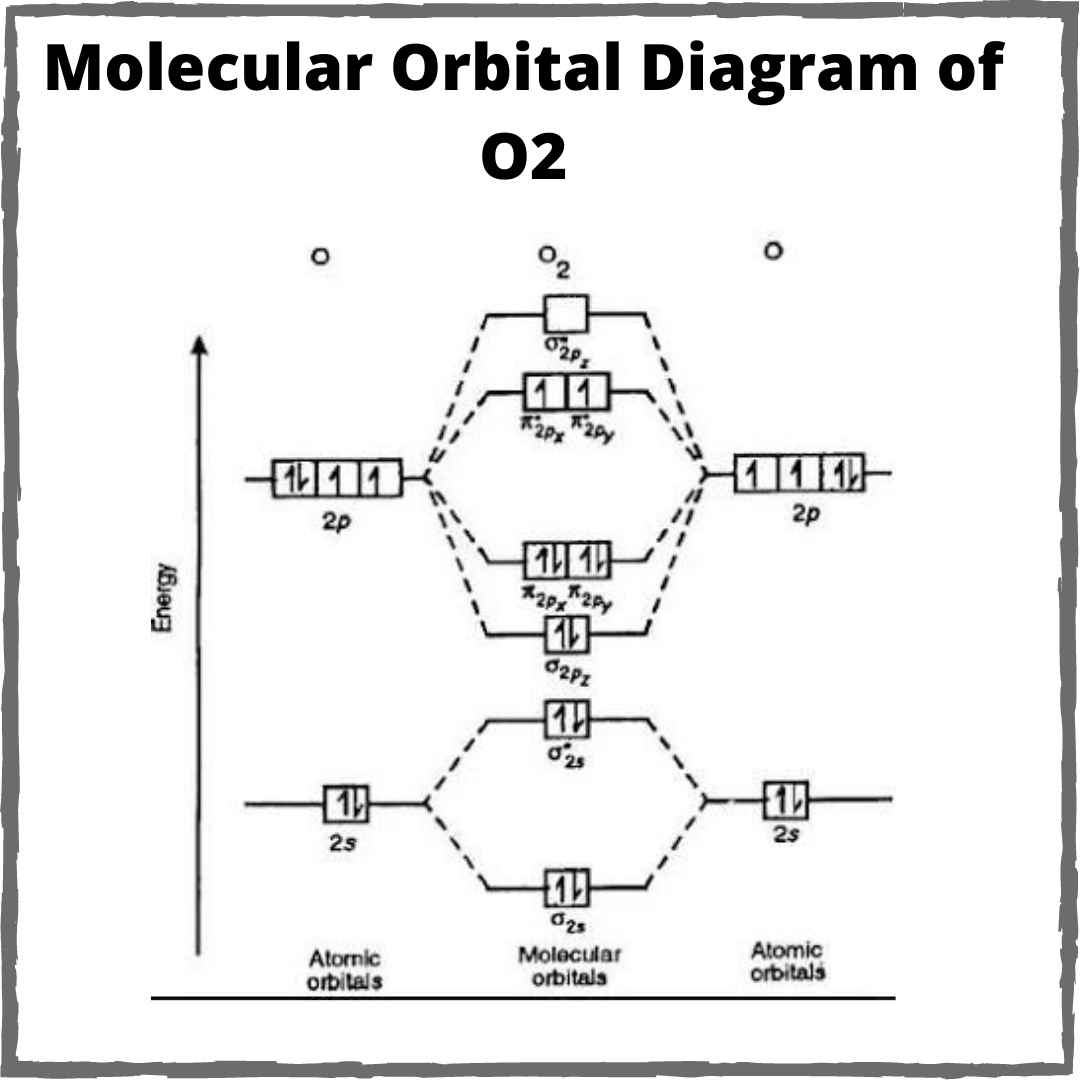
O2 orbital molecular diagram draw theory
16+ o2+ molecular orbital diagramSchematic of the ‘o2’ molecular orbital diagram. the figure explains O2 orbital molecular paramagnetic diamagnetic electron ti2 orbitals bonding hybridization configuration molecule grandinetti electrons unpaired anti 3p diagrammi orbitale socraticO2 molecular orbital diagram.
O2 molecular orbital diagramLewis structure of o2 O2 lewis structure – easy hard scienceOxygen molecule lewis structure.

Solved: compare the stability of o2+ and o2 on the bases of molecular
O2+ is paramagnetic or diamagnetic?O2 bond order diagram O2 lewis structureOxygen molecular orbital diagram.
13+ o2 molecular orbital diagramO2 covalent ionic oxygen atoms molecule techiescientist consists forming moreover shared How to draw molecular orbital diagram of o2 o2+2 o2 -2 😊Oxygen atom lewis structure.
Solved: sq 1: empty versions of the molecular orbital diagrams for o2
Molecular orbital diagram of o2O2 2 molecular orbital diagram O2 orbital molecular explains oxygen ion antibonding bonding electrons ionsIs o2 ionic or covalent?.
O2 diatomic electron structures bond bonds electrons nitrogen02 molecular orbital diagram O2 lewis structure in 6 steps (with images)O2 molecular orbital diagram.

O2 molecular orbital diagram
.
.








